 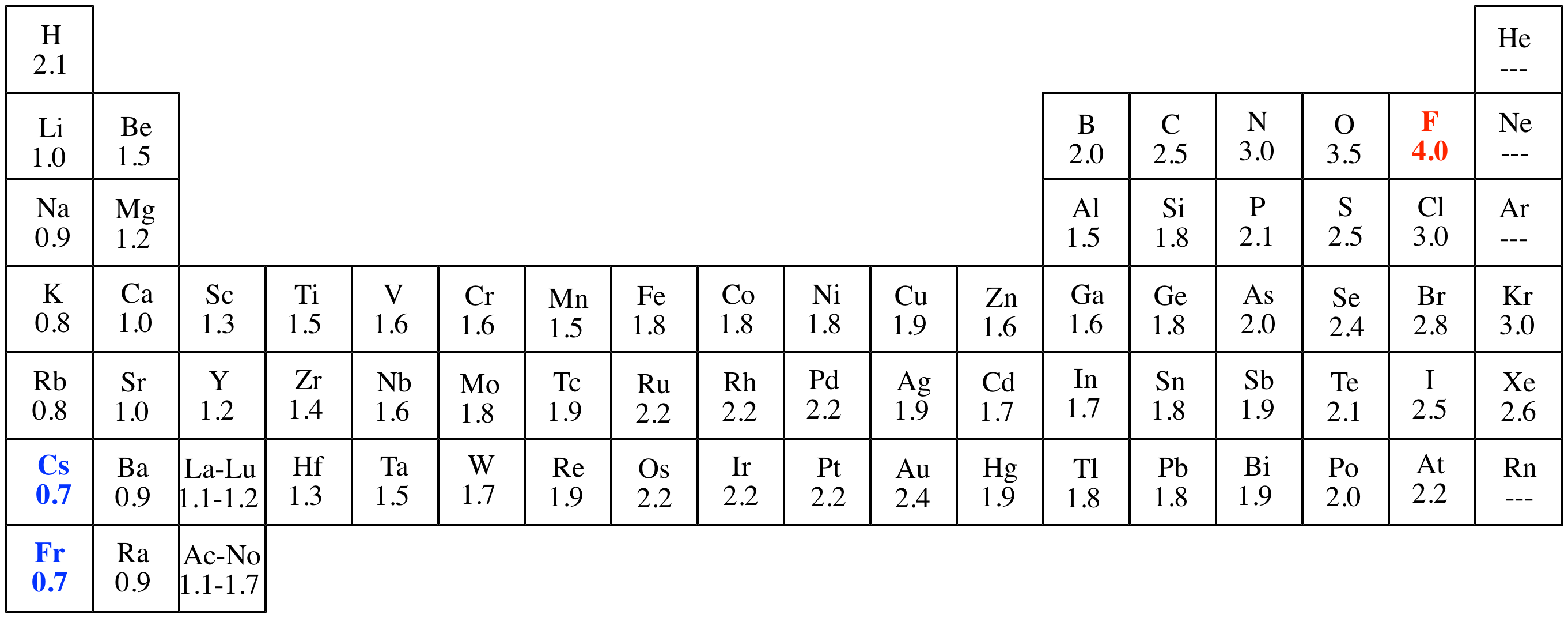
Gas which are stable and don’t need any electrons) will have gained one or two electrons and become a negative ion (anion) because it now has At the same time the atom from the right side of the Periodic Table (but not a noble Protons than electrons and will have a positive charge. Each element becomes an ion.Īn atom from the left side of the Periodic Table tends to lose an electron thereby becoming a positive ion (cation) because it now has more Some elements on the other hand will seek the stable configuration by moving electrons from one atom to another. When two atoms have equal electronegativity they bond covalently, that is they share the electrons. Have no electronegativity because they don’t form bonds. Fluorine is considered to have the highest electronegativity. So as you can see elements on the left hand (low electronegativity) of the Period Table of Elements, those in columns 1 and 2 have one or two moreĮlectrons than those in the most stable column 18 (the noble gases). The noble gasesĪnd are considered inert, that is they have low reactivity. Remember: when the highest occupied energy level of an atom is filled with electrons, the atom is stable and not likely to react. 
Pauling’s is the most commonly used scale for describing an atom’s electronegativity, hydrogen has a very low electronegativity and chlorine has a very high one. Remember that your textbook publisher may have help links as well.ĭefinition: Electronegativity is an atomic property which has to do with the tendency to attract the electrons of another atom to form anĭiscoverer: Linus Pauling defined this concept and the “Pauling” scale was named for him. Links to helpful pages will also be at the end. On this page you will find information related to electronegativty in physical chemistry. The Periodic Chart of Table of the ElementsĮlectronegativity Written by tutor Kathie Z.Explanation of States of Matter Problems.SN1SN2 – Nucleophilic Substitution Reactions.Explanation of Numbers and Math Problems – Set 3.Explanation of Numbers and Math Problems – Set 2.Explanation of Numbers and Math Problems – Set 1.Metric Prefixes and Their Origins and Use.Drawing Cyclohexane Rings – Organic Chemistry.Alkanes and Alkenes – Organic Chemistry.Light & Dark Reactions in Photosynthesis.Search Lessons Search for: Science Resources and Science Lessons – Science Help 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
